Boc SPPS possesses a number of advantages compared to Fmoc SPPS: higher solubility of Boc amino acids promotes faster coupling; TFA fully solvates the peptide–resin at each deprotection cycle, preventing peptide–resin aggregation and enabling the synthesis of long peptides; in comparison to Fmoc SPPS, there is much less aspartimide formation. In addition Boc deprotection is always complete, in contrast to Fmoc where partial deprotection during aggregation is problematic. Because of these attendant benefits a combination of Boc SPPS and native chemical ligation (NCL) has enabled the synthesis of many proteins of sufficient quality to be crystallized for structural studies. Peptide thioesters were prepared directly by tert‐butyloxycarbonyl (Boc) solid phase peptide synthesis (SPPS) facilitated by the stability of the thioester bond to trifluoroacetic acid (TFA), used for Boc deprotection cycles. The major limitation of Boc SPPS is its requirement for anhydrous HF for the deprotection and cleavage of the peptide from the resin. HF requires specialized apparatus and training.
Showing 1–36 of 114 resultsSorted by latest
-

4,4′-Bis(2-bromoacetyl)biphenyl
$ 250.0 Add to cart -

Boc-L-Proline
$ 120.0 Add to cart -

Boc-D-Phenylglycinol
$ 150.0 Add to cart -

Boc-D-β-HoSer(OBzl)-OH
$ 250.0 Add to cart -

Boc-β-HoSer(OBzl)-OH
$ 180.0 Add to cart -
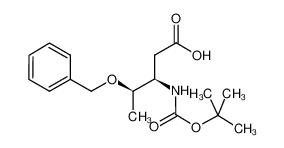
Boc-β-HoThr(OBzl)-OH
$ 300.0 Add to cart -
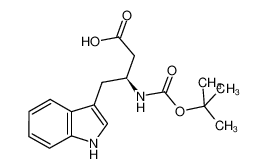
Boc-β-HoTrp-OH
$ 200.0 Add to cart -

Boc-β-HoTyr(OBzl)-OH
$ 200.0 Add to cart -
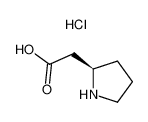
Boc-D-β-Homoproline
$ 750.0 Add to cart -
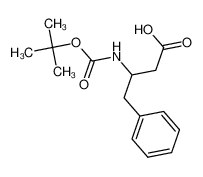
Boc-DL-β-Homophenylalanine
$ 750.0 Add to cart -
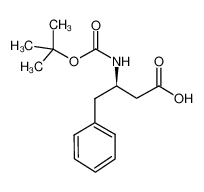
Boc-D-β-HoPhe-OH
$ 750.0 Add to cart -
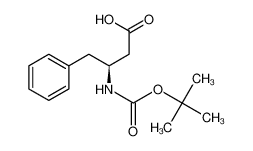
Boc-β-HoPhe-OH
$ 750.0 Add to cart -
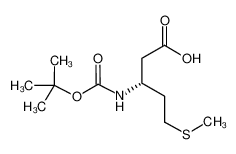
Boc-L-β-Homo-Met-OH
$ 750.0 Add to cart -

Boc-L-β-Homo-Lys(Cbz)-OH
$ 1,250.0 Add to cart -
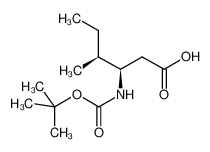
Boc-L-β-Homo-Ile-OH
$ 1,250.0 Add to cart -

Boc-L-β-Homo-Gln-OH
$ 250.0 Add to cart -

Boc-β-HoGlu(OBzl)-OH
$ 1,250.0 Add to cart -
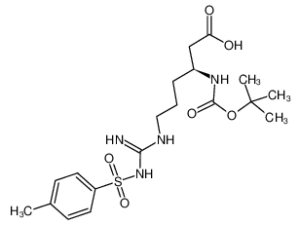
Boc-L-β-Homo-Arg(Tos)-OH
$ 1,250.0 Add to cart -
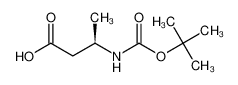
Boc-D-β-HoAla-OH
$ 350.0 Add to cart -
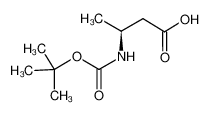
Boc-L-β-HoAla-OH
$ 200.0 Add to cart -

Boc-Cys(SO3H)-OH·disodium salt
$ 3,500.0 Add to cart -
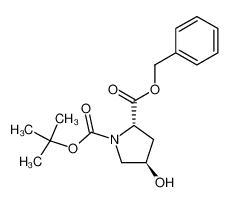
Boc-Hyp-OBzl
$ 450.0 Add to cart -
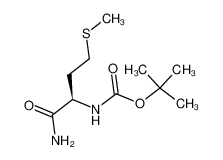
Boc-D-Met-NH2
$ 450.0 Add to cart -

Boc-D-Pyr-OBzl
$ 275.0 Add to cart -
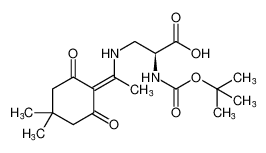
Boc-Dap(Dde)-OH
$ 225.0 Add to cart -
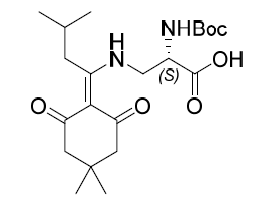
Boc-Dap(ivDde)-OH
$ 450.0 Add to cart -

Boc-D-β-Phe(3-Cl)-OH
$ 280.0 Add to cart -

Boc-diethanolamine
Read more -

Boc-Ser(BzPEG1)-OH
$ 480.0 Add to cart -
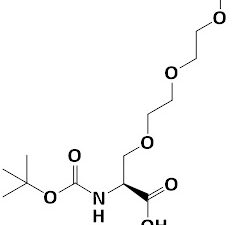
Boc-Ser(mPEG2)-OH
$ 800.0 Add to cart -
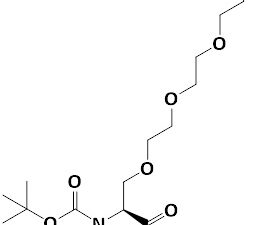
Boc-Ser(Azido-PEG3)-OH
$ 1,000.0 Add to cart -

Boc-Ser(mPEG1)-OH
$ 300.0 Add to cart -
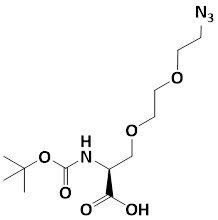
Boc-Ser(azido-PEG2)-OH
$ 700.0 Add to cart -
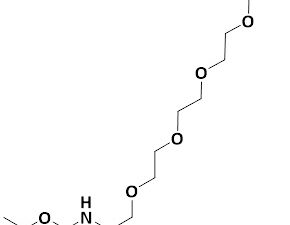
Boc-Ser(azido-PEG4)-OH
$ 800.0 Add to cart -
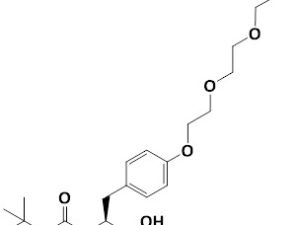
Boc-Tyr(PEG3)-OH
$ 480.0 Add to cart -
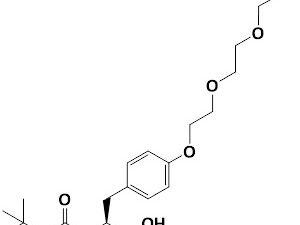
Boc-Tyr(azido-PEG3)-OH
$ 800.0 Add to cart